Biomed Diagnostics
Saline Solution 0.85% Media Tubes, pack/50
- Part Number:
- BIOM-11-612-002
- Lead Time:
- Call for current availability
- Shipping:
- FREE SHIPPING on most orders over $50*
- Quantity:
- each
- Style:
- cell culture
- Country of Origin:
- Made in the USA
Description
 Saline (0.85%) Viral Transport Solution for Nucleic Acid Testing of SARS-CoV-2. This contains an isotonic solution typically used for detection of the severe acute respiratory syndrome SARS-CoV-2.
Saline (0.85%) Viral Transport Solution for Nucleic Acid Testing of SARS-CoV-2. This contains an isotonic solution typically used for detection of the severe acute respiratory syndrome SARS-CoV-2.
- To be used with Nasopharyngeal (NP) or Oropharyngeal (OP) specimens.
- Room Temperature Shelf Life: 60 days
- Cured, distilled, de-mineralized salt water solution
- Product is packed in 50-count boxes (#11-612-002)
- Custom batch pours are 3000 vials each
- Lead time 2 weeks.
- Blanket POs accepted for regularly scheduled shipments
- FDA / CDC approved
- Refrigerate at 2-8 degrees C after inoculation
- Download the Saline Solution information sheet (PDF)
Saline Solution (0.85%)
The Saline Solution (0.85%) is an isotonic solution, manufactured in compliance with ISO 13485:2016 standards. It is intended to be inoculated with nasopharyngeal (NP) or oropharyngeal (OP) synthetic fiber swab specimens (Not provided, see IFU for details), to be analyzed in the laboratory with validated qRT-PCR assays. This Saline Solution (0.85%) has been validated for the detection of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that causes COVID-19 disease in humans.1,2 The use of this device for other viral specimens is the responsibility of the end user including the validation thereof.
Specifications
| Product Principle |
|
|---|---|
| Simple Convenience |
|
| Storage |
Do not store frozen. Keep away from direct light and heat exposure. Do not use expired solution. Do not use |
| Specimen |
Nasopharyngeal (NP) or oropharyngeal (OP) synthetic fiber swab specimens |
Performance Characteristics This test was performed to evaluate the Biomed Saline Solution (0.85%) by detection of heat inactivated cell lysate from SARS-Cov-2 (ATCC® VR-1986HK™) infected cells, after RNA was isolated using the QIAamp® Viral RNA Mini Kit (QIAGEN®). Detection of the isolated SARSCov-2 RNA was by qRT-PCR using New England Biolabs® OneTaq® One-Step RT-PCR kit and run on a Roche® Lightcycler® 96 with EvaGreen® (Biotium®) detection from samples after storage of viral lysates in Saline Solution (0.85%) at 24, 48 and 72 hours at room temperature (25°C). Synthetic RNA standard (BEI Resources) at 2.9 x 108 was serially (10-fold) diluted for the standard (STD) in the amplification reaction. The assay results demonstrate consistent amplification and Cq quantification of the nCov-2 N gene (using the N1 CDC primer) in the solution after incubations. In addition, excellent dilution linearity is demonstrated across all samples and replicates (See Figure 1) indicating consistency in the performance of the solution Saline Solution (0.85%) replicate samples and incubation time. This data indicates that the Biomed ISO 13845 manufactured Saline Solution (0.85%) does not negatively interfere with the qRT-PCR detection of Sars-Cov-2 viral nucleic acid materials after incubation at room temperature 25°C for 72 hours. A delay in amplification signal was observed after 168-hour (7 days) incubation at 25°C, probably due to specimen degradation. However, incubation at 4°C for 168 hours (7 days) did not negatively interfere with the qRT-PCR performance. In conclusion, the result indicates that the Saline Solution (0.85%) is compatible with the designated viral specimen inoculation, nucleic acid extraction and qRT–PCR assay when used as described herein.
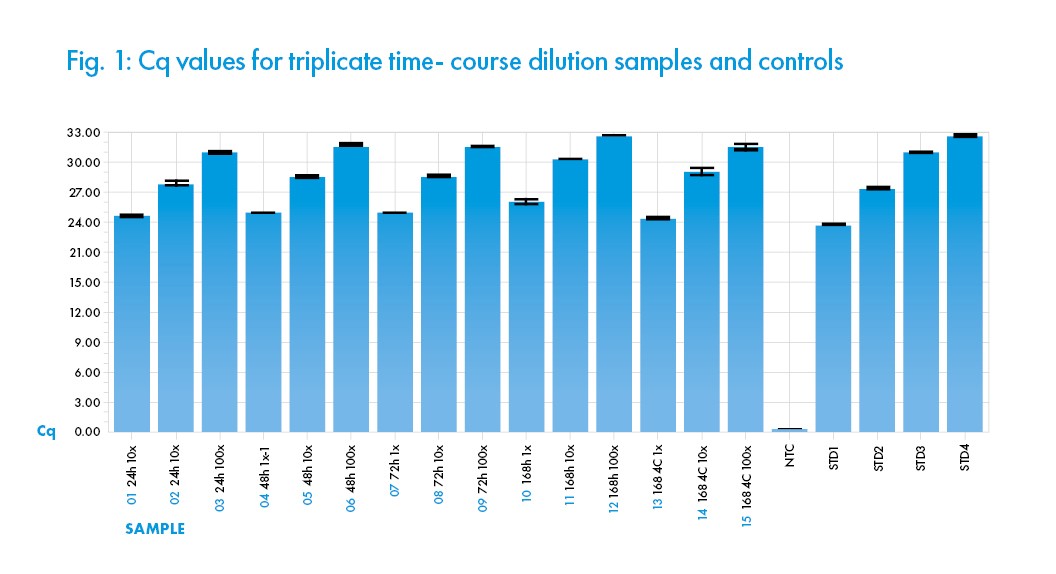 All samples shown are means from triplicate assays, with error bars indicating the arithmetic error for Cq shown. The performance of the qRT-PCR reaction assessed with standard curves indicated R2 of 0.98, Efficiency of 114%, and Slope of -3.033. All the values fall within acceptable quantitative PCR ranges.
All samples shown are means from triplicate assays, with error bars indicating the arithmetic error for Cq shown. The performance of the qRT-PCR reaction assessed with standard curves indicated R2 of 0.98, Efficiency of 114%, and Slope of -3.033. All the values fall within acceptable quantitative PCR ranges.
Regulatory
For in vitro diagnostic use.
Not available in all countries; please inquire.
FDA listed
References
- FAQs on Viral Transport Media During COVID-19
- CDC preventing transmission of infectious agents in healthcare settings guidelines
Intended Use
Saline Solution (0.85%) is a saline solution for viral specimens.
Description and Principle
The Saline Solution (0.85%) is an isotonic solution, manufactured in compliance with ISO 13485:2016 standards. It is intended to be inoculated with nasopharyngeal (NP) or oropharyngeal (OP) synthetic fiber swab specimens (Not provided, see “Procedure” section for details), to be analyzed in the laboratory with validated qRT-PCR assays.
This Saline Solution (0.85%) has been validated for the detection of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that causes COVID-19 disease in humans.1,2 The use of this device for other viral specimens is the responsibility of the end user including the validation thereof. When used according to the instructions for use, the Saline Solution (0.85%) ensures a non-replicating competent status of SARS-CoV-2 (COVID-19) thereby preserving viral RNA genome integrity of the virus.
The Saline Solution (0.85%) is aseptic and does not contain bacteria or fungi growth-inhibition agents. Therefore, specimen should be handled under suitable conditions that do not encourage growth of these microbes. From the site of collection to downstream laboratory nucleic acid testing (e.g., qRT-PCR), the Saline Solution (0.85%) is intended to aid in the process of collection and transport of human clinical specimens that contain the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It is the responsibility of the end user to validate the use of this device for viral specimens other than described here.
The Saline Solution (0.85%) is validated to be compatible with the identification of SARS-CoV-2 using qRT-PCR technology by:
- Aiding the safe transport and preservation of the specimen.
- Viral specimen stored in the Saline Solution (0.85%) at room temperature (18–25 °C) for 72 hours is compatible with downstream qRT-PCR tests.
- Compatible with approved nucleic acid extraction and qRT-PCR tests.
Reagents and Appearance
The Saline Solution (0.85%) in polypropylene tubes appears clear and contains 8.5 g Sodium Chloride per liter of deionized water. Post-production pH of the solution is 6.5 ± 1.0 at 25°C.
Precautions, Safety and Disposal
Read the Safety Data Sheet (SDS) and follow the handling instructions. Wear appropriate protective eyewear, clothing and gloves. Do not use if package is damaged or leaking. All specimens should be handled according to the CDC preventing transmission of infectious agents in healthcare settings guidelines2 cdc.gov/infectioncontrol/guidelines/isolation/index.html Once the tube has been inoculated and resealed, re-open only in a biological safety cabinet. Prior to disposal, sterilize tubes by autoclaving at 121°C for 20 minutes or through another suitable means of sterilization. Any serious incident that occurs in relation to this device shall be reported to the manufacturer and the competent authority, as required, of the country in which the user and/or the patient is established.
Storage
Do not store frozen. Keep away from direct light and heat exposure. Do not use expired solution. Do not use the solution if it appears to be damaged, leaking or the solution appears to be cloudy or evaporated.








